 On Oct. 26, 2005, Alfred Caronia, a sales consultant for a little-known pharmaceutical company based in California, met with a doctor to discuss promotion of one of the firm’s drugs.
On Oct. 26, 2005, Alfred Caronia, a sales consultant for a little-known pharmaceutical company based in California, met with a doctor to discuss promotion of one of the firm’s drugs.
The drug, a depressant called Xyrem, had been approved by the U.S. Food and Drug Administration to treat only certain patients with the sleep disorder narcolepsy. But Caronia maintained, in a conversation with a doctor that was recorded by federal investigators, that the drug could be used to treat an array of other sleep and muscle ailments.
There was a problem with Caronia’s claims: None of those extra uses for Xyrem, the prescription version of a “date rape” street drug, had been approved by the FDA. In the eyes of the agency, the benefits of the unapproved uses were unproven and the results potentially dangerous.
In 2008, Caronia was convicted of violating federal law by pushing Xyrem for unapproved purposes, a practice known as off-label promotion. But in 2012, a federal appeals court, in a 2-to-1 decision, threw out Caronia’s conviction on grounds that had nothing to do with the safety of the drug or its potential misuse. The court found that “the government prosecuted Caronia for mere off-label promotion,” and that such activity is protected commercial speech under the First Amendment, as long as it not shown to be false or misleading.
The case smoldered for several years, but is now increasingly being recognized as a potential landmark decision that could have profound implications for the FDA and public health.
Two settlements since late 2015 apparently influenced by the Caronia ruling, and a case pending before another federal appeals court, are fueling debate about whether the First Amendment’s protection of commercial speech could undermine the FDA’s ability to prevent drug marketing abuses.
What’s more, changing political dynamics — especially the anti-regulatory aims of the Trump administration and Republican-dominated Congress — could free drug makers to push the boundaries of the law.
The stakes are high. Off-label promotion has long been considered a serious white-collar crime. The FDA has fined drug companies billions of dollars for off-label violations. The aim has been to prevent them from overstating the benefits and understating the risks of their products, a practice known as misbranding — and one that sometimes leads to patients getting drugs that prove harmful or even deadly.
The trend toward curtailing off-labeling restrictions worries Aaron Kesselheim, a physician and lawyer at Harvard University and an expert on drug and medical device policy issues. “Pharmaceutical manufacturers have a long history of making promotional statements around biased studies, or emphasizing the positive studies and deemphasizing the negative studies,” Kesselheim said.
“When they’ve done that in the past, it’s led to substantial morbidity and mortality because of the power of manufacturers’ promotional measures in influencing physicians’ prescribing methods.”
As an example of past drug company abuses, Kesselheim cited the promotion of antipsychotic medicines to elderly patients, which he said were associated with a 60 percent to 70 percent increase in death rate, and the promotion of antidepressants to children, which were linked to an increase in suicidal behavior.
Federal prosecutors haven’t quit pressing ahead with off-label marketing lawsuits. In July, for example, Celgene Corp., a drug company based in Summit, N.J., agreed to pay $280 million to settle fraud allegations linked to the promotion of two cancer treatment drugs for uses not approved by the FDA. Unlike the Caronia dispute, though, the case focused not on free speech issues, but with allegations the company violated the federal False Claims Act by submitting false claims to Medicare, and to 28 states and the District of Columbia.
The Caronia decision meshes neatly with the Trump administration’s anti-regulatory agenda. On Jan. 31, just days after his inauguration, President Trump delivered a welcome message to a gathering of pharmaceutical industry executives that he would be “streamlining” the FDA’s approval process. “We’re going to get rid of a tremendous number of regulations,” he said, according to a White House press release.
Trump in March also pleased industry executives with his choice to head the FDA, Scott Gottlieb, a physician who favors allowing the promotion of unapproved uses for drugs. “The sharing of scientific evidence can sometimes have important public health benefits,” he wrote in a 2008 commentary. He chastised those who “pursue a rigid adherence to restrictions” on drug promotion.
The FDA turned down a request for an interview with Gottlieb. The agency, which held a little-noted meeting on off-label promotion last November just two days after the presidential election, said it is “engaged in a comprehensive review of its regulations and policies” regarding off-label communications. The outcome is being eagerly awaited by the Pharmaceutical Research and Manufacturers of America, the dominant pharmaceutical industry trade group. It said at the meeting that its members are asking “that FDA establish a clear, safe harbor” for drug makers to “share truthful and non-misleading information” about off-label uses.
* * *
Under federal law, the FDA approves drugs for particular uses when drug makers submit scientific evidence demonstrating that those uses are safe and effective. At that point the companies are allowed to promote the drugs for those specific uses.
With doctors, the situation is different. The FDA says it is not eager to interfere with “the practice of medicine,” so it has allowed doctors the flexibility to prescribe drugs for unapproved uses. Doctors and researchers can also share information about unapproved uses at medical meetings or in the pages of medical journals. Part of the reasoning in the 2012 Caronia ruling was that, if doctors are going prescribe off-label uses anyway, shouldn’t all of the truthful information about the drugs be available to them?
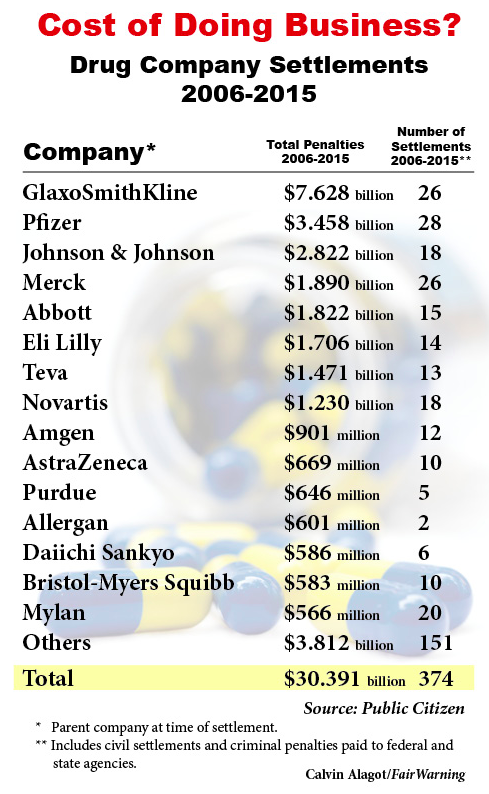
Drug companies that violate the law sometimes have paid a steep price. The pharmaceutical industry paid more than $30 billion in fines between 2006 and 2015 for off-label promotion and other offenses. At the top of the list is pharmaceutical giant GlaxoSmithKline. In 2012, it pleaded guilty and paid $3 billion to settle criminal and civil charges related to the off-label promotion of the antidepressants Paxil and Wellbutrin, and the diabetes drug Avandia.
The Justice Department said the company, among other things, sponsored “dinner programs, lunch programs, spa programs and similar activities to promote the use of Paxil in children and adolescents” — even though the anti-depressant hadn’t been approved for use in patients under 18. The Justice Department also said Glaxo prepared a misleading medical article portraying Paxil as effective, while suppressing two studies that found that it wasn’t.
* * *
The drug at the center of the Caronia case, Xyrem, won FDA approval in 2002. It was marketed by Orphan Medical Inc., a small company in Minnetonka, Minn., that specialized in so-called orphan drugs. Such drugs treat rare diseases and, as a result, often bring in little revenue and get scant attention from drug marketers or researchers. By 2005, Xyrem was generating $20 million in revenue, pocket change in an industry where blockbuster drugs can yield annual sales in the billions.
Xyrem’s prospects seemed limited in part because the FDA required every package to carry a “black box” warning, the agency’s most serious advisory for prescription drugs. The current wording of the warning, in an apparent reference to the drug’s link to date rape, cites the risks of “misuse and abuse,” and noted that the “adverse reactions” associated with Xyrem’s active ingredient have included “decreased consciousness, coma and death.” The active ingredient, often called GHB, can “make you become weak and confused — or even pass out — so you can’t tell if you are being drugged,” says the federal government’s Office of Women’s Health.
The FDA also required Orphan Medical to establish a tightly regulated distribution and education network for Xyrem.
Yet Orphan Medical — and later on its new owner, Jazz Pharmaceuticals Inc., which moved its headquarters from Palo Alto, Calif., to Dublin, Ireland, in 2012 — had ambitious plans. Alfred Caronia was hired to promote Xyrem in and around the New York City area as part of an aggressive marketing effort.
Caronia’s charge was to boost sales and make Xyrem much more than a niche drug. His approach was revealed in the October 2005 taped conversation with a doctor who was serving as a government informant in an investigation of Orphan Medical. Caronia suggested unapproved uses of the medicine; he also told the doctor about the diagnostic codes to use to get insurance reimbursement. And Caronia encouraged the doctor to prescribe it for children and elderly patients, for whom it had not been approved. According to the recording, Caronia said: “There have been reports of patients as young as fourteen using it and obviously greater than sixty-five,” Caronia said. “It’s a very safe drug.”
Drug-enforcement officials did not see it that way. On the streets, Xyrem was known as GHB, or liquid ecstasy. It became popular in the 1990s at “raves” in which it was used to boost libido and to cause amnesia, leaving users vulnerable to sexual assault. Its sometimes deadly side effects include difficulty breathing while asleep, depression, nausea, vomiting, seizures and comas.
Investigators eventually zeroed in on Caronia. In July, 2007, a grand jury returned an indictment charging him with two misdemeanor offenses related to introducing a misbranded drug into interstate commerce. (Another Jazz Pharmaceuticals consultant was also indicted in connection with running a speakers’ program promoting Xyrem.) In October, 2007, Caronia sought to have the charges dismissed on the grounds that it restricted his First Amendment free speech rights and that the misbranding provisions were unconstitutionally “vague and broad.”
In October, 2008, a jury convicted Caronia “of conspiring to introduce a prescription drug into interstate commerce with the intent that it be used in ways its labeling neither disclosed nor described.” Just over a year later, he was sentenced to one year of probation, 100 hours of community service, and a small fine. He appealed.
In December, 2012, the Second Circuit Court of Appeals in New York, in a 2-to-1 decision, vacated the conviction on free-speech grounds. “We do not hold, of course, that the FDA cannot regulate the marketing of prescription drugs,” the court said. “We conclude simply that the government cannot prosecute pharmaceutical manufacturers and their representatives . . . for speech promoting the lawful, off-label use of an FDA-approved drug.”
But the dissenting judge, Judge Debra Ann Livingston, wrote that “the majority calls into question the very foundations of our century-old system of drug regulation,” leaving companies with little incentive to seek approval for off-label uses of a drug.
Caronia was fired by Jazz, has left the pharmaceutical industry, and is now working as a carpenter, according to his attorney in the case, Thomas Liotti of Garden City, N.Y. Caronia “didn’t do anything wrong here,” said Liotti. “He presented the warning labels to the doctors.” The doctor who cooperated with federal investigators “had a script prepared by FDA agents” to get Caronia “to say this, or get him to say that,” Liotti said.
* * *
The Caronia case is now beginning to cause some ripples. “Caronia upended decades of pharmaceutical law,” Christopher Robertson, a University of Arizona’s law professor, wrote in June in a discussion paper called “The Tip of the Iceberg.” He noted that the FDA did not appeal the decision, maintaining that the ruling would have little effect on other cases.
But recent history has proven otherwise. Last year the FDA agreed to a settlement that allows Amarin Corp. to engage in truthful promotion of its fish oil drug Vascepa for unapproved uses. And it allows the company to affirm whether the promotion meets the standard.
In 2012, Amarin won FDA approval for Vascepa to treat high levels of certain fats in the blood. In the wake of the decision, the company sought permission from the FDA to promote the drug for use in patients with lower levels of the fats, called triglycerides.
The FDA refused, and Amarin sued, making a First Amendment argument and invoking the Caronia case as a precedent. In August, 2015, a federal judge in Manhattan agreed with the company, ruling that under Caronia, the FDA can’t accuse a company of misbranding “based on truthful promotional speech alone.”
In March, 2016, the FDA settled the case with Amarin and said it would allow the expanded promotion “based on scientific evidence that was previously submitted.” That outcome demonstrated that the agency “has departed significantly from its traditional position on the issue,” policy analysts Deborah Mazer and Gregory Curfman wrote on the blog of the journal Health Affairs. They added that “its potential reach is vast.”
In December, 2015, while the Amarin battle was under way, the FDA settled with another company that raised a First Amendment argument, Pacira. The company won the ability to promote the expanded use of its pain drug Exparel. The FDA rescinded a warning letter it sent the company the year before noting that the drug was approved by the agency only for treatment of pain after surgery for bunions or hemorrhoids.
Meanwhile, another new legal battle with free speech implications has come up in the First Circuit Court of Appeals in Boston. It is the first off-label promotion case outside the Second Circuit, where the others arose. It involves a medical device to clear sinuses, made by Acclarent. Two executives convicted of marketing the device for off-label uses are appealing.
Congress has also shown interest in loosening restrictions. On July 12, while efforts to repeal and replace Obamacare were dominating the headlines, the House Subcommittee on Health held a hearing to review two bills relating to promotion of off-label uses of drugs and devices.
The subcommittee, chaired by Texas Republican Michael C. Burgess, a doctor, said the idea behind the bills was to spread scientific information to help doctors make the right decisions on use of drug. “A lack of relevant information can lead to physicians making patient care decisions with incomplete information. This is both unfair to the physician and unsafe for the patient.”
Oregon Republican Greg Walden, chairman of the Energy and Commerce Committee, of which the health subcommittee is a part, said the bills “do not provide manufacturers with free rein to communicate any and all information about their products,” but they do allow drug companies to “responsibly disseminate accurate, and up-to-date information about medical products.”
In a background memo prepared for the hearing, the subcommittee said that proposed legislation would clarify what drug makers can say without fear of retribution — namely, they must be clear about the data supporting their claims and not claim that unapproved uses are safe and effective.
But R. Alta Charo, a professor of law and bioethics at the University of Wisconsin, raised warning flags. In prepared testimony for the hearing, she said the bill “risks eviscerating the general rule against off-label promotion,” with potential harm to patients. “Studies have repeatedly shown that even products that look promising in early trials will usually be shown to be unsafe or ineffective when larger trials are completed.”
Other observers, such as the University of Arizona’s Robertson, view the push toward greater latitude for off-label promotion as part of a broader legal pattern, including U.S. Supreme Court rulings striking down laws on First Amendment grounds. He pointed to the Citizens United case that expanded free speech protections to corporations, and a ruling that overturned a law banning depictions of dog fighting. Robertson also cited the Hobby Lobby decision, which recognized the religious freedom rights of business owners. “In almost every way you can imagine, they are breaking down the wall over what is protected speech, what isn’t. And in every case, it’s always more power to the First Amendment,” he said.


