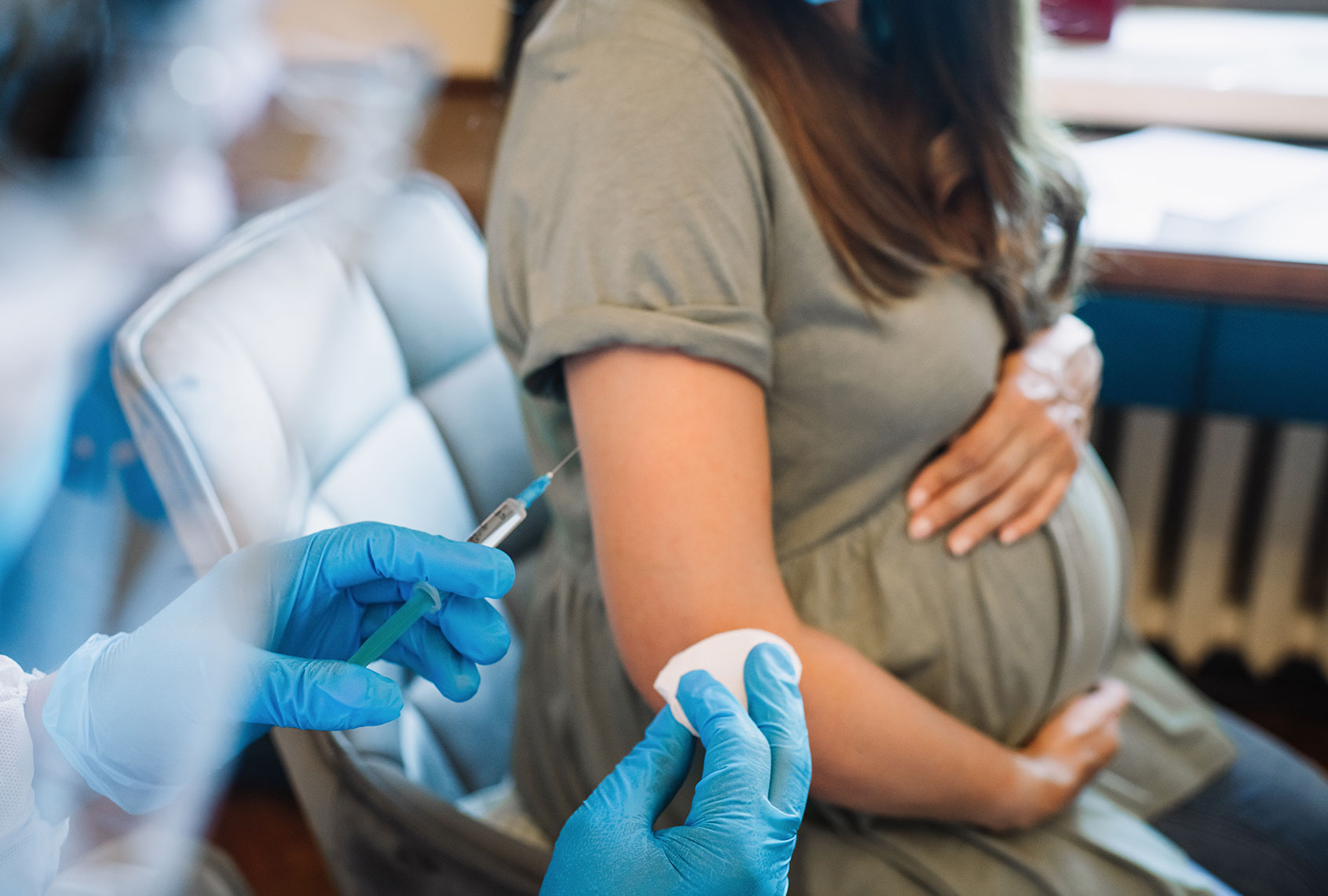
Kia Slade was seven months pregnant, unvaccinated, and fighting for breath, her oxygen levels plummeting, when her son came into the world last May.
A severe case of covid pneumonia had left Slade delirious. When the intensive care team tried to place an oxygen mask on her face, she snatched it away, she recalled. Her baby’s heart rate began to drop.
Slade’s doctor performed an emergency cesarean section at her bedside in the intensive care unit, delivering baby Tristan 10 weeks early. He weighed just 2 pounds, 14 ounces, about half the size of small full-term baby.
But Slade wouldn’t meet him until July. She was on a ventilator in a medically induced coma for eight weeks, and she developed a serious infection and blood clot while unconscious. It was only after a perilous 2½ months in the hospital, during which her heart stopped twice, that Slade was vaccinated against covid-19.
“I wish I had gotten the vaccine earlier,” said Slade, 42, who remains too sick to return to work as a special education teacher in Baltimore. Doctors “kept pushing me to get vaccinated, but there just wasn’t enough information out there for me to do it.”
A year ago, there was little to no vaccine safety data for pregnant people like Slade, because they had been excluded from clinical trials run by Pfizer, Moderna, and other vaccine makers.
Lacking data, health experts were unsure and divided about how to advise expectant parents. Although U.S. health officials permitted pregnant people to be vaccinated, the World Health Organization in January 2021 actually discouraged them from doing so; it later reversed that recommendation.
The uncertainty led many women to delay vaccination, and only about two-thirds of the pregnant people who have been tracked by the Centers for Disease Control and Prevention were fully vaccinated as of Feb. 5, leaving many expectant moms at a high risk of infection and life-threatening complications.
More than 29,000 pregnant people have been hospitalized with covid and 274 have died, according to the CDC.
“There were surely women who were hospitalized because there wasn’t information available to them,” said Dr. Paul Offit, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia.
Vaccine developers say that pregnant people — who have special health needs and risks — were excluded from clinical trials to protect them from potential side effects of novel technologies, including the Pfizer and Moderna mRNA vaccines and formulations made with cold viruses, such as the Johnson & Johnson vaccine.
But a KHN analysis also shows that pregnant people were left behind because including them in vaccine studies would have complicated and potentially delayed the delivery of covid vaccines to the broader population.
A growing number of women’s health researchers and advocates say that excluding pregnant people — and the months-long delay in recommending that they be immunized — helped fuel widespread vaccine hesitancy in this vulnerable group.
“Women and their unborn fetuses are dying of covid infection,” said Dr. Jane Van Dis, an OB-GYN at the University of Rochester Medical Center who has treated many patients like Slade. “Our failure as a society to vaccinate women in pregnancy will be remembered by the children and families who lost their mothers to this disease.”
New Technology, Uncertain Risks
At the time covid vaccines were being developed, scientists had very little experience using mRNA vaccines in pregnant women, said Dr. Jacqueline Miller, a senior vice president involved in vaccine research at Moderna.
“When you study anything in pregnant women, you have two patients, the mom and the unborn child,” Miller said. “Until we had more safety data on the platform, it wasn’t something we wanted to undertake.”
But Offit notes that vaccines have a strong record of safety in pregnancy and sees no reason to have excluded pregnant people. None of the vaccines currently in use — including the chickenpox and rubella vaccines, which contain live viruses — have been shown to harm fetuses, he said. Doctors routinely recommend that pregnant people receive pertussis and flu vaccinations.
Offit, the co-inventor of a rotavirus vaccine, said that some concerns about vaccines stem from commercial, not medical, interests. Drugmakers don’t want to risk that their product will be blamed for any problems occurring in pregnant people, even if coincidental, he said.
“These companies don’t want bad news,” Offit said.
In the United States, health officials typically would have told expectant mothers not to take a vaccine that was untested during pregnancy, said Offit, a member of a committee that advises the FDA on vaccines.
Due to the urgency of the pandemic, health agencies instead permitted pregnant people to make up their own minds about vaccines without recommending them.
Women’s medical associations were also hampered by the lack of data. Neither the American College of Obstetricians and Gynecologists nor the Society for Maternal-Fetal Medicine actively encouraged pregnant people to be vaccinated until July 30, after the first real-world vaccine studies had been published. The CDC followed suit in August.
“If we had had this data in the beginning, we would have been able to vaccinate more women,” said Dr. Kelli Burroughs, the department chairman of obstetrics and gynecology at Memorial Hermann Sugar Land Hospital near Houston.
Yet anti-vaccine groups wasted no time in scaring pregnant people, flooding social media with misinformation about impaired fertility and harm to the fetus.
In the first few months after the covid vaccines were approved, some doctors were ambivalent about recommending them, and some still advise pregnant patients against vaccination.
An estimated 67% of pregnant people today are fully vaccinated, compared with about 89% of people 65 and older, another high-risk group, and 65% of Americans overall. Vaccination rates are lower among minorities, with 65% of expectant Hispanic mothers and 53% of pregnant African Americans fully vaccinated, according to the CDC.
Vaccination is especially important during pregnancy, due to increased risks of hospitalization, ICU admission and mechanical ventilation, Burroughs said. A study released in February from the National Institutes of Health found that pregnant people with a moderate to severe covid infection also were more likely to have a C-section, deliver preterm, or develop a postpartum hemorrhage.
Black moms such as Slade were already at higher risk of maternal and infant mortality before the pandemic, due to higher underlying risks, unequal access to health care, and other factors. Covid has only magnified those risks, said Burroughs, who has persuaded reluctant patients by revealing that she had a healthy pregnancy and child after being vaccinated.
Slade said she has never opposed vaccines and had no hesitation about receiving other vaccines while pregnant. But she said she “just wasn’t comfortable” with covid shots.
“If there had been data out there saying the covid shot was safe, and that nothing would happen to my baby and there was no risk of birth defects, I would have taken it,” said Slade, who has had Type 2 diabetes for 12 years.
Working at Warp Speed
Government scientists at the NIH were concerned about the risk of covid to pregnant people from the very beginning and knew that expectant moms needed vaccines as much or more than anyone else, said Dr. Larry Corey, a leader of the COVID-19 Prevention Network, which coordinated covid vaccine trials for the federal government.
But including pregnant volunteers in the larger vaccine trials could have led to interruptions and delays, Corey said. Researchers would have had to enroll thousands of pregnant volunteers to achieve statistically robust results that weren’t due to chance, he said.
Pregnancy can bring on a wide range of complications: gestational diabetes, hypertension, anemia, bleeding, blood clots, or problems with the placenta, for example. Up to 20% of people who know they’re pregnant miscarry. Because researchers would have been obliged to investigate any medical problem to make sure it wasn’t caused by one of the covid vaccines, including pregnant people might have meant having to hit pause on those trials, Corey said.
With death tolls from the pandemic mounting, “we had a mission to do this as quickly and as thoroughly as possible,” Corey said. Making covid vaccines available within a year “saved hundreds of thousands of lives.”
The first data on covid vaccine safety in pregnancy was published in April, when the CDC released an analysis of nearly 36,000 vaccinated pregnant people who had enrolled in a registry called V-safe, which allows users to log the dates of their vaccinations and any subsequent symptoms.
Later research showed that covid vaccines weren’t associated with increased risk of miscarriage or premature delivery.
Dr. Brenna Hughes, a maternal-fetal medicine specialist and member of the American College of Obstetricians and Gynecologists’ covid expert group, agrees that adding pregnant people to large-scale covid vaccine and drug trials may have been impractical. But researchers could have launched parallel trials of pregnant women, once early studies showed the vaccines were safe in humans, she said.
“Would it have been hard? Everything with covid is hard,” Hughes said. “But it would have been feasible.”
The FDA requires that researchers perform additional animal studies — called developmental and reproductive toxicity studies — before testing vaccines in pregnant people. Although these studies are essential, they take five to six months, and weren’t completed until late 2020, around the time the first covid vaccines were authorized for adults, said Dr. Emily Erbelding, director of microbiology and infectious diseases at the National Institute of Allergy and Infectious Diseases, part of NIH.
Pregnancy studies “were an afterthought,” said Dr. Irina Burd, director of Johns Hopkins’ Integrated Research Center for Fetal Medicine and a professor of gynecology and obstetrics. “They should have been done sooner.”
The NIH is conducting a study of pregnant and postpartum people who decided on their own to be vaccinated, Erbelding said. The study is due to be completed by July 2023.
Janssen and Moderna are also conducting studies in pregnant people, both due to be completed in 2024.
Pfizer scientists encountered problems when they initiated a clinical trial, which would have randomly assigned pregnant people to receive either a vaccine or placebo. Once vaccines were widely available, many patients weren’t willing to take a chance on being unvaccinated until after delivery.
Pfizer has stopped recruiting patients and has not said whether it will publicly report any data from the trial.
Hughes said vaccine developers need to include pregnant people from the very beginning.
“There is this notion of protecting pregnant people from research,” Hughes said. “But we should be protecting patients through research, not from research.”
Recovering Physically and Emotionally
Slade still regrets being deprived of time with her children while she fought the disease.
Being on a ventilator kept her from spending those early weeks with her newborn, or from seeing her 9-year-old daughter, Zoe.
Even when Slade was finally able to see her son, she wasn’t able to tell him she loved him or sing a lullaby, or even talk at all, due to a breathing tube in her throat.
Today, Slade is a strong advocate of covid vaccinations, urging her friends and family to get their shots to avoid suffering the way she has.
Slade had to relearn to walk after being bedridden for weeks. Her many weeks on a ventilator may have contributed to her stomach paralysis, which often causes intense pain, nausea and even vomiting when she eats or drinks. Slade weighs 50 pounds less today than before she became pregnant and has resorted to going to the emergency room when the pain is unbearable. “Most days, I’m just miserable,” Slade said.
Her family suffered, as well. Like many babies born prematurely, Tristan, now nearly 9 months old and crawling, receives physical therapy to strengthen his muscles. At 15 pounds, Tristan is largely healthy, although his doctor said he has symptoms of asthma.
Slade said she would like to attend family counseling with Zoe, who rarely complains and tends to keep her feelings to herself. Slade knows her illness must have been terrifying for her little girl.
“The other day she was talking to me,” Slade said, “and she said, ‘You know, I almost had to bury you.”