For parents, recent revelations regarding the ultra-contagiousness of the coronavirus’ delta variant could not have come at a worse time.
As parents gear up to send their children back to school for what many hoped would finally be a “normal” school year, news of the rapidly spreading delta variant have made “normal” seem much further off. From rising cases across the country to new masking guidance from the U.S. Centers for Disease Control and Prevention (CDC), there is a lot for parents to navigate as schools reopen this August and September.
Unlike last year at this time, kids over the age of 12 can get vaccinated before returning to school, which will increase the chances of stopping transmission in classroom settings in high schools. That should give some comfort to parents of teenagers.
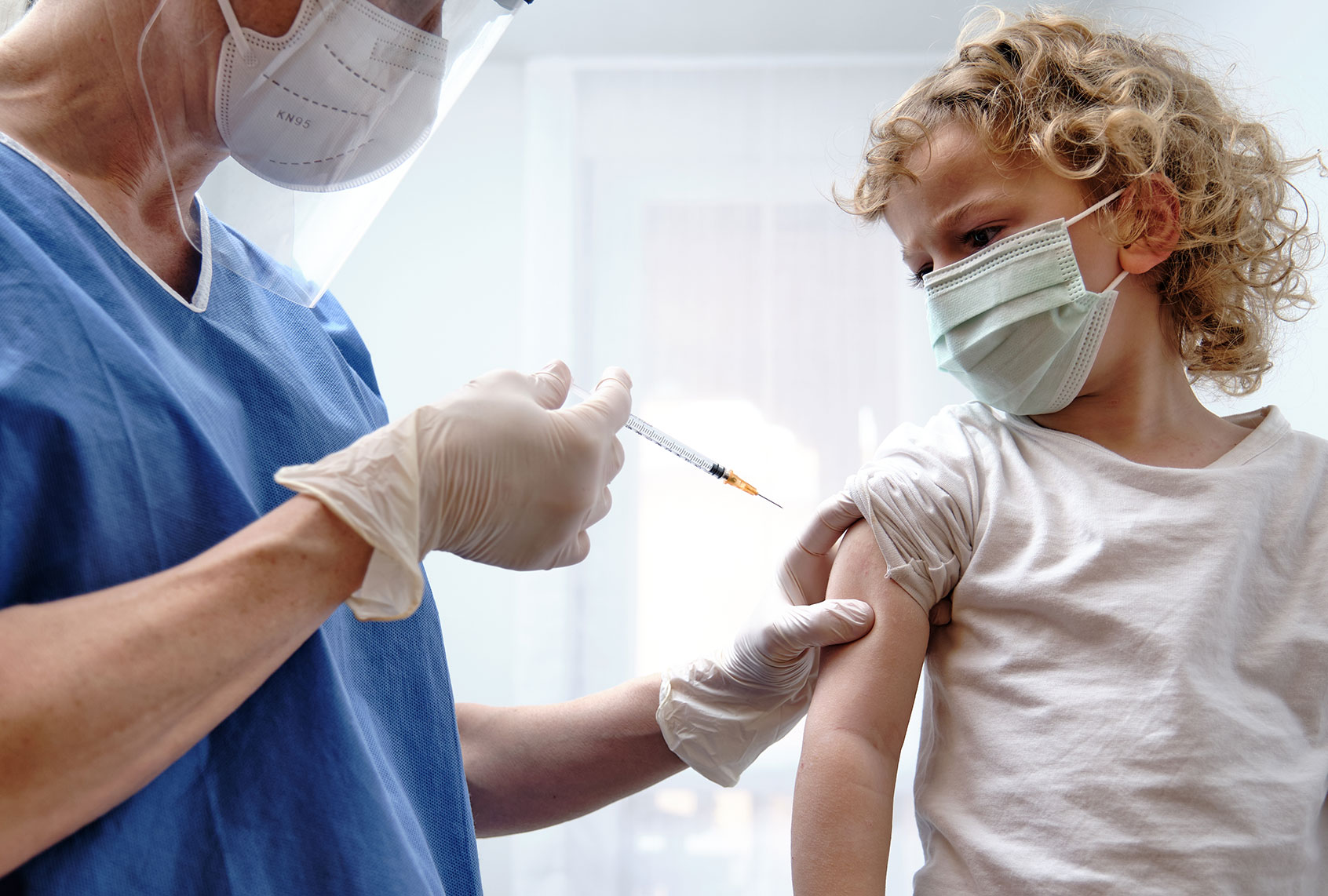
But for children under twelve, it’s a different story. There are no approved vaccines for them, and while infected children are not generally known to get severe cases of COVID-19 (though it does happen sometimes), they can still spread the virus to adults in their vicinity.
Yet after a year and half of navigating the pandemic, schools know that coronavirus mitigation methods — such as wearing face masks, social distancing, contact tracing and monitoring symptoms in students — can reduce the likelihood of transmission in a school setting. However, the most effective way to contain this virus, even if breakthrough infections with the delta variant can happen, is through mass vaccination.
So how much longer will parents with children under the age of 12 have to wait?
In an emailed statement to Salon, a spokesperson for Pfizer said a phase 2-3 study began in June 2021 “to further evaluate the safety, tolerability and immunogenicity of [vaccine] BNT162b2 in preventing COVID-19 in healthy children between the ages of 6 months to 11 years old.” That means it may only be a matter of time until a vaccine for the younger set is ready.
[Dig deeper: Here’s what the different “phases” of vaccine production really mean]
“The companies [Pfizer and BioNTech] expect to have the safety and immunogenicity data that could potentially support an Emergency Use Authorization (EUA) for use in children ages 5 to 11 years old, if such an EUA is deemed necessary, by the end of September,” the statement said. “The full dataset from this study, which will be required to support licensure in this age group, is expected by the end of 2021; similar data packages will be submitted shortly thereafter to support EUA and licensure in children 6 months to 5 years of age.”
In other words, late September is the earliest parents of 5 to 11 year olds could expect their children to be eligible for vaccination. Currently, Pfizer is on a faster track than Moderna.
However, last week federal regulators requested that the vaccine companies expand their trials to test coronavirus vaccines in several thousand school-aged children before requesting EUA. The request was made to assess whether or not a rare side effect, myocarditis — which is inflammation of the heart — is more common in children. According to the Washington Post, this might cause vaccine authorization for 5 through 11-year-olds to be delayed to late October or early November. In turn, that could mean that children under the age of 5 won’t be eligible until 2022.
As Salon has previously explained, it is standard practice to test older children first, because children of different ages can have a different response to the vaccine. The goal of clinical trials with children is to find a balance between the correct age and dosage of the vaccine in which a strong immune response is triggered without too many side effects. The variables in a clinical trial with children are different.
“Children’s immune systems are different — they’ve had prior exposures, their immune systems may not be as experienced, and children also weigh less than older individuals,” said Dean Blumberg, chief of pediatric infectious diseases and associate professor in the Department of Pediatrics at the University of California, Davis. “Getting the dose, right, is important, and we know that with some vaccines, what you need to do is give a higher dose of the vaccine in younger children because they haven’t been exposed to the antigen, the active component of the vaccine previously, and in other cases you give a lower dose, because it’s more weight-based.”
Want more health and science stories in your inbox? Subscribe to Salon’s weekly newsletter The Vulgar Scientist.
Blumberg said in children over the age of 12, Pfizer and Moderna use the same dose as in adults. However, in younger children, the dose is lower.
As Blumberg mentioned, one potential side effect on the FDA’s radar is myocarditis. According to the CDC as of July 26, 2021, Vaccine Adverse Event Reporting System (VAERS) has received 1,194 reports of myocarditis or pericarditis among people under the age of 30; most reports have been in male adolescents.
While this might be alarming, Blumberg emphasized the importance of weighing the risks and benefits.
“We know that Myocarditis occurs following infection, too — and if you look at the numbers, the risk of myocarditis is much higher following natural infection than following vaccination,” Blumberg said. “So the benefits outweigh the risks. But you have to sort those things out and get the data, and that’s why they have to study it.”
From a public health standpoint, the race is on to increase vaccine accessibility to children. That’s because the more children that get vaccinated, the less likely the coronavirus can mutate into something even more dangerous than delta.
“Delta variant is likely not the last variant that we’re going to see,” Blumberg said. “The virus can further evolve, and there’s greater chances of further evolution when it multiplies out of control and into unvaccinated populations.”
This is partly why it’s important for schools to keep transmission low, which can be done by following proper mitigation strategies among unvaccinated people. Teachers’ unions have been especially vocal about the need for school staff and students to be vaccinated in order to promote the safety of all.
“We continue to urge educators, school staff and eligible students to get vaccinated, and for our schools to serve as community vaccination sites,” said California Teachers Association President E. Toby Boyd in a statement last week. “Vaccines, along with multiple layers of safety protections – including masks, testing and physical distancing – are our primary and most important defense against COVID and are key to ensuring safe in-person instruction can continue this fall.”